Recce Pharmaceuticals Secures China Patent for Lead Anti-Infectives, Strengthening Global IP Strategy
Recce Pharmaceuticals secures Chinese patent acceptance for its flagship anti-infective compounds, expanding its global IP footprint into the world’s second-largest pharmaceutical market.

( Image: ACB News)
█ ACB News Cathy
SYDNEY, 27 May 2025 – Recce Pharmaceuticals Ltd (ASX: RCE), an Australian biotech developing next-generation synthetic anti-infectives, has received official notification from the China National Intellectual Property Administration (CNIPA) confirming the acceptance of its core Patent Family 4, valid until 2041.
The newly accepted patent covers RECCE® 327 and RECCE® 529, including their manufacturing processes, therapeutic applications for bacterial and viral infections, and multiple delivery formats (oral, injection, transdermal, aerosol, etc.).
This is Recce’s fifth Patent Family 4 grant, following approvals in Australia, Canada, Israel, and Japan. Additional international applications under the Patent Cooperation Treaty (PCT) are under review.
CEO James Graham welcomed the decision, stating it marks a significant expansion of Recce’s global IP portfolio into China, the world’s second-largest pharmaceutical market, where the antibiotic sector alone is valued at US$4.09 billion and forecast to grow at a 5.7% CAGR through 2030.
Recce’s pipeline includes:
● R327: IV/topical therapy for drug-resistant bacterial infections
● R435: Oral antibacterial
● R529: Antiviral therapy
All three compounds have been recognised by the WHO and the FDA, with R327 receiving QIDP status, Fast Track designation, and 10-year post-approval market exclusivity.
Recce owns and operates its own automated manufacturing facility in Australia, supporting current clinical trials and future commercial supply.
This patent marks a key step in Recce’s global commercialisation strategy and its efforts to combat the global threat of antimicrobial resistance.
Copyright Notice:
ACB News reserves full copyright over all articles explicitly marked as "original" content.
No media outlet, corporate website, platform, or app within Australia may reproduce, excerpt, adapt, or republish such content in any form without prior written authorization from ACB News.
Any unauthorized use or reproduction—including via third-party platforms—will be considered an infringement. ACB News reserves the right to pursue legal action against any such violations.
相关阅读

-
【异动股】Recce Pharmaceuticals(ASX:RCE)与美国陆军签订战伤护理抗感染研究协议
专注于新一代合成抗感染药物的澳大利亚生物医药企业 Recce Pharmaceuticals Ltd (ASX股票代码:RCE) 宣布与美国陆军外科研究所(USAISR)签署合作研发协议,以评估RECCE 327凝胶在烧伤伤口感染中的应用效果。

-
Recce Pharmaceuticals中国专利获批 拓展全球抗感染新药知识产权布局
Recce首席执行官 James Graham 表示:“感谢中国国家知识产权局对Recce抗感染技术优势的认可。此次专利获批标志着我们全球知识产权组合的重要扩展,覆盖至全球第二大医药市场。”

-
TELIX Pharmaceuticals (ASX:TLX) 前列腺癌成像剂新药申请获中国药品监管机构受理
TELIX Pharmaceuticals Ltd (ASX股票代码:TLX)向市场披露,中国药品监管机构已受理其主打药品前列腺癌成像剂TLX591-Px(商品名为Illuccix)的新药申请。

-
【异动股】癌症药预测效果获临床前验证 AI药物发现平台Algorae Pharmaceuticals(ASX:1AI)股价节节飙升
伴随多项积极运营进展的披露,AI赋能制药公司Algorae Pharmaceuticals Ltd (ASX股票代码:1AI) 股价节节攀升,近一月累计上涨超88%。
-
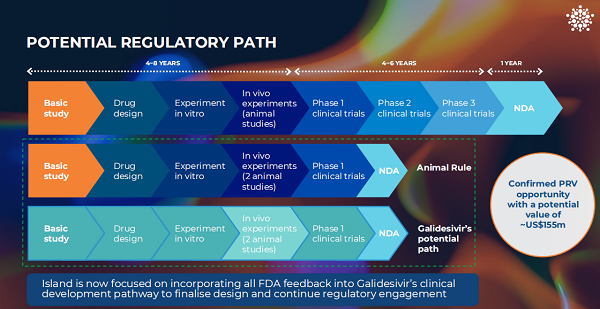
【异动股】Island Pharmaceuticals (ASX:ILA)获FDA批准采用动物规则途径并取得热带病优先审评券
经美国FDA回复确认,Island Pharmaceuticals Ltd (ASX股票代码:ILA) 可按照动物规则途径推进使用加德西韦(Galidesivir)治疗马尔堡病毒的药物审批,该药获批后Island还将有资格获得热带病优先审评券(PRV)。
-
【异动股】Island Pharmaceuticals (ASX:ILA) 股价持续飙升:加速推进马尔堡病毒治疗药FDA审批进程
从事抗病毒药物开发的Island Pharmaceuticals Ltd (ASX股票代码:ILA)延续强劲涨势,自发布季报以来过去一周上涨19.74%,目前已较7月初的低点上扬230%。

-
Neuren Pharmaceuticals(ASX: NEU)2025财年上半年: 利润稳健增长产品管线持续推进 国际化布局加速
Neuren Pharmaceuticals(ASX代码:NEU)周三发布2025财年上半年财报,公司依托核心产品DAYBUE™(trofinetide)授权收入实现税后净利润1,500万澳元,同比增长87.5%。同时,第二款候选药物NNZ-2591在多项罕见神经发育障碍适应症的临床开发进展顺利,持续巩固Neuren在罕见病治疗领域的领先地位。

-
Neuren Pharmaceuticals(ASX:NEU)启动全球首个PMS三期临床试验
致力于开发针对儿童早发严重神经系统疾病新型药物的澳交所上市公司Neuren Pharmaceuticals(ASX代码:NEU)本周一宣布,其治疗费朗-麦克德米德综合征(Phelan-McDermid综合征 ,PMS)候选药物 NNZ-2591 的三期临床试验,已在美国首个研究中心启动,获得伦理审查委员会(IRB)批准,其他试验点也在陆续推进中。
免责声明:本网站信息仅供一般参考,不构成投资或财务建议。虽力求准确与完整,但不保证信息的准确性、完整性或时效性。投资有风险,决策前请咨询专业独立顾问。使用本网站即视为接受本免责声明。
热门点击
-
- 澳财长发布临时指令 禁止香港股东转让重稀土公司Northern Minerals(ASX:NTU)股份或行使AGM表决权
-
- 联邦银行罕见暴跌折射财案深层次冲击 税改撼动盈利基石 大银行股市场主导地位或将松动
-
- 澳预算案CGT改革落地 成长股或将遭弃 高股息价值股迎布局良机
-
- 项目融资临近落地 610万盎司金矿开发迎关键拐点 Theta Gold Mines(ASX:TGM)迈入加速兑现阶段
-
- 【5.18】今日财经时讯及重要市场资讯
-
- 澳财长依据外资审查法签发新处置令 强制Northern Minerals(ASX:NTU)六家中资关联股东剥离股份
-
- 【5.19】今日财经时讯及重要市场资讯
-
- 联邦银行罕见暴跌折射财案深层次冲击 税改撼动盈利基石 大银行股市场主导地位或将松动
-
- 【新股跟踪】国防无人机制造配套供应商KTEK Aerosystems Ltd (ASX股票代码:KTK) 登陆ASX 首秀暴涨102%
-
- 利率上调叠加税改担忧 澳洲房拍清盘率遭遇寒流 潜在买家驻足观望
-
- 中石油转让Browse气田合资项目权益 战略性撤离澳洲长期上游资产
-
- 【异动股】Hawk Resources (ASX:HWK)单周上涨逾三成 启动近600万澳元融资 战略聚焦Olympus钪矿及美国铜资产勘探
-
- 【异动股】农企巨头Elders (ASX:ELD) 重跌22%:并购整合期遭受财务阵痛 净债务攀升杠杆率骤涨
-
- 澳洲女首富Gina Rinehart斥资近亿美元加仓美国军工股 持续重仓稀土矿业板块
-
- 【5.20】今日财经时讯及重要市场资讯



